The percentage of s character in sp, sp2, and sp3 hybridized carbon is 50%, 33.33%, and 25%, respectively. In ethyne, each carbon atom is sp-hybridized. In this way, four sp-orbital are generated. One sp-orbital of each carbon atom by overlapping forms a sigma bond between carbon atoms. Remaining one sp-orbital of each carbon atom overlap with 1s-orbital of hydrogen atom to produce two sigma bonds.
What is an sp2 hybridized carbon atom?
Count the sigma bonds. How to clean android phone storage. Subatomic. All carbon atoms (with a very few exceptions) involved in an organic molecule will have 4 bonds. But not all of them are sigma bonds. A sigma bond is the primary bond, as opposed to a pi bond, which is the second or third bo. Sp2hybridisation When a C atom is attached to 3 groups and so is involved in 3 σ bonds, it requires 3 orbitals in the hybrid set. This requires that it is sp2hybridised. The general 'steps' are similar to that.
1 Answer
carbon with #sp^2# hybridized atomic orbital is formed by mixing one s and two p atomic orbitals.
Explanation:
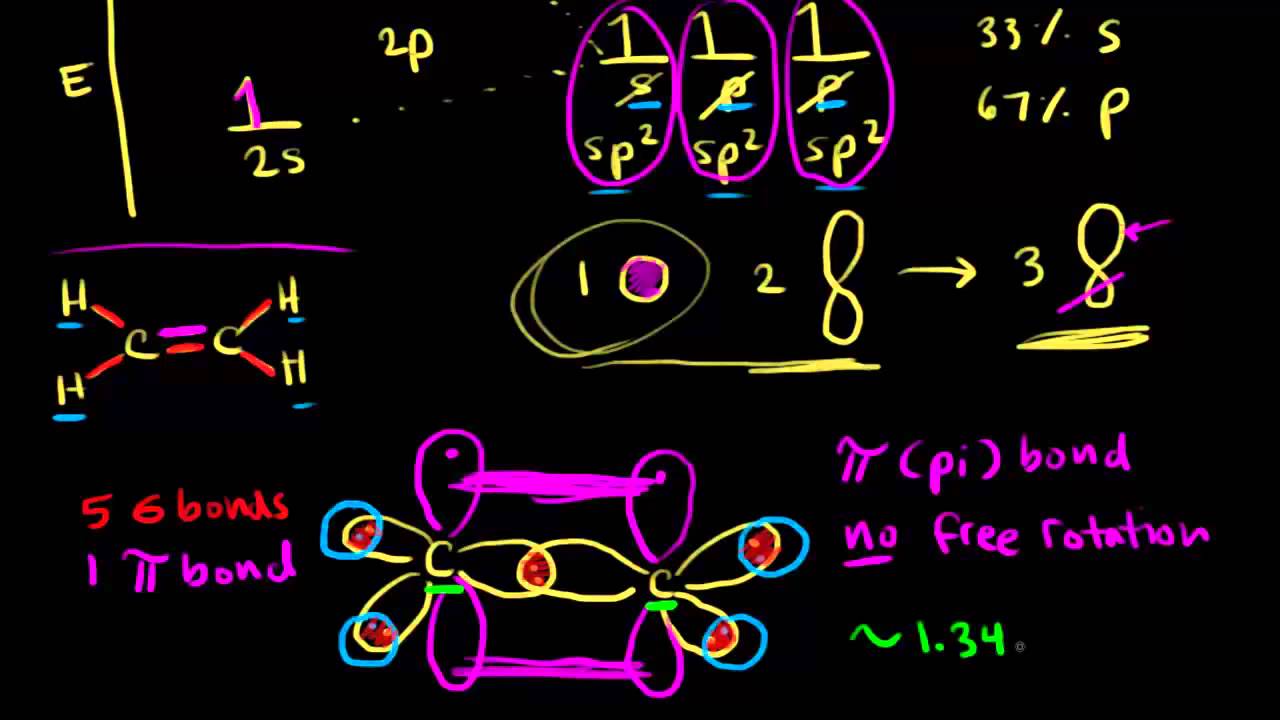
An example of carbon with #sp^2# hybridized atomic orbital is alkene, specifically the two carbons involved in the C=C. Each of that carbon has 3 sigma bonds and 1 pi bond.
Carbon is tetravalent (forms 4 bond) and the ground state electron configuration cannot explain its valency since there's only 2 unpaired electron (left image below).
Sp2 Carbon Ir
Therefore, one of the electron in 2s will be promoted to the empty 2pz orbital (middle image below). How to clear cache internal storage android.
In order to form the 3 sigma bonds in ethene, one 2s and two of the 2p orbitals will mix to form three#2sp^2# hybridized orbitals (right image below). These three hybridized orbital will bond with other atoms to form sigma bonds. The remaining one 2p orbital will form a pi bond with 2p orbital of the other carbon through sideway overlap.
Note that the three#2sp^2# hybridized orbitals are all degenerate and have lower energy compared to 2p orbital. They will arrange themselves into trigonal planar geometry. Also, they have 33% s character [1/3*100 = 33%].
